A Tale of Two Biopsies: Liquid Biopsy vs Tissue Biopsy
The Power of Liquid Biopsy
Liquid biopsy is the innovative solution to cancer detection and treatment. While liquid biopsies are revolutionizing oncology, they are not yet ready to entirely replace their competition, solid tissue biopsies. This article examines the differences between the two forms of biopsy, and how both must be used together for the most effective outcomes.
Extraction Methods of Biopsies
The initial component in both of these biopsy methods is the extraction of matter to be analyzed. (Right: Comparison of Biopsy Methods Gen News ).
Liquid biopsies work by measuring cell-free DNA (cfDNA) in a sample of fluid from the body. It is theorized that when cell death occurs, the cell releases DNA into the fluids they are associated with. In oncology, some of the most important cfDNA are circulating tumor DNA (ctDNA), which come from a cancerous cell. Also significant for analysis are circulating tumor cells (CTC), which shed from a primary tumor.
Liquid biopsies often collect fluid samples in traditional non-invasive ways; blood samples are drawn intravenously, urine is collected in a sterile cup, and saliva is swabbed in the mouth. While all of these methods are considered less intrusive compared to tissue biopsies, cerebrospinal fluid (CSF) is extracted using a spinal tap in a relatively painful procedure. When a liquid biopsy is performed as a screening tool, it is possible that all of the cells will be analyzed to find any markers that match diseases. In oncology, the liquid sample is analyzed specifically to find mutations for cancer.
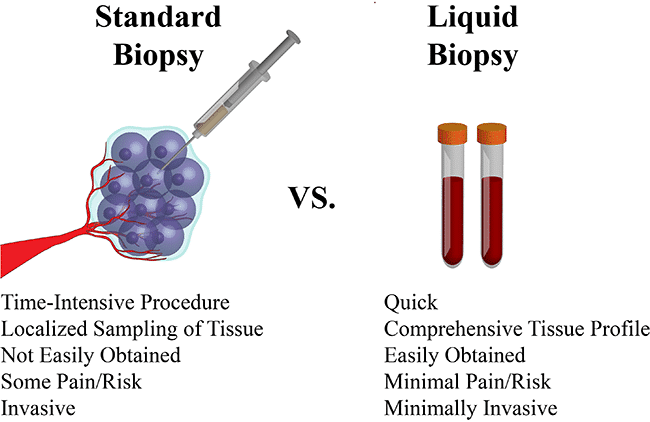
Tissue biopsies require solid matter from the body. This specimen is removed either directly from a tumor or from bone marrow. An anesthetic may be used: local to numb area of extraction for pain, or general to make the patient unconscious. Techniques can involve needles, endoscopic tools, or excision using a sharp utensil to cut tissue. Specimen extraction from the body may require surgical methods. As a result, side effects can include risk of infection, internal bleeding, or prolonged recovery time.
Once extracted, the tumor specimen is then preserved and analyzed for mutations. An unfortunate challenge in tissue that has not been preserved is tissue necrosis, which occurs quickly in tumor cells (Parnell). Unfortunately, preservation methods involve the use of chemicals that may degrade the effectiveness of a sample. This is the case of formalin fixation, which displays higher levels of a certain allele frequency range and results in false positives for cancer diagnosis (Ilie & Hoffman).
Comparing Functions in Each Biopsy
Tissue or liquid sample collected in a biopsy is tested for cancer by analyzing genetic material. A common method of analysis is whole genome sequencing (WGS), which maps an organism’s genome and tracks specific mutations associated with cancer. Research has sequenced exons, or sections of DNA, and found markers associated with types of cancer, such as the protein HER2 in breast cancer (Shtivelman).
Liquid biopsies utilize genetic sequencing for many purposes in oncology. They can be uniquely used as a screening tool for early detection either by testing the entire sample for mutations, or looking for specific mutations in the sample. Furthermore, liquid biopsies are vital in precision treatment and tracking relapse in patients with remission (Gen News). In all of these cases, WGS methods are used to detect genetic alterations of interest. Liquid testing also uses a much smaller sample and has a significantly quicker turnaround for results compared to tissue biopsies, partly due to WGS automation processes.

Another area where liquid biopsies excel is when a tumor has metastasized, meaning it has spread to multiple sites in the body (Parnell). It is not practical, or often possible, to collect tissue from each tumor for analysis. Thus, liquid biopsies are more advantageous to prove an overview of the mutations present in each of the tumor sites, or when tumors are in an unreachable area where tissue cannot be collected via needle or surgical methods and cfDNA must be used.
A drawback is that cfDNA in liquid is extremely limited and detection sensitivity is low. For example, a study on prostate cancer reported that two separate liquid biopsy companies found matching mutations for only 7.5% of patients (Mone). This indicates that certain mutations may be under or over represented in a random sample. CTCs or ctDNA may be from a source that is no longer a threat, or the genes present in them are from a tiny fraction of the tumor. Alternately, the sample may not represent mutations that actually are present in a tumor.
Tissue biopsies are irreplaceable in identifying the nature of tumors: type of cancer, gene expression of the tumor, and presence of treatment-resistant mutations. However, that is affected by several components of origin, such as tumor location, tissue amount available, and accessibility for surgical removal. The instable nature of tumors makes them heterogeneous, which means tissue from a certain area of the tumor may have radically different mutations and genetic expressions than other areas (Shtivelman). This provides inaccurate information.
Deciding Which Biopsy to Use
Research demonstrates that neither biopsy method is capable of sampling an entire tumor and revealing complete details of all present mutations. This is a setback for both diagnosis and treatment, which tend to be based around the mutations.
Tissue biopsies are fully utilized when a known tumor’s location is confirmed and available for extraction. Liquid biopsies are best used for screening, identifying mutations in metastatic cancer, and tracking changes in mutations for treatment (Zhang). Both biopsy methods can be used for tracking changes during treatment, but liquid biopsies are more sustainable for patient health. While the biopsies may have their strengths, it is important to note how they differ.
Several comparison studies reveal discrepancies in data. In one study, tissue and liquid biopsies were done simultaneously for various types of cancer. The results found 45 total mutations, but the overlap of those found by both tests was only 10, or 22% (Kuderer et al.)
In general practice, this would determine precision treatment by drugs, but only 9 out of 36 recommended drugs were concordant, or 25%. Another similar study tracking specific genetic mutations found consistent results in only 17% of cases (Chae, et al.)
While these results point to the necessity of using both biopsy methods, tissue biopsies provide more accurate results in some cases. In a study done on pancreatic cancer, tissue biopsies found relevant gene variants in 87% of instances, compared to 25% found by liquid biopsies (Harris). Additionally, liquid biopsies missed the most common pancreatic cancer mutation in 29% of opportunities, compared to 13% missed in tissue biopsies.
At the moment, liquid and tissue biopsies function as two distinct tests with different methods and functions. Using both biopsy methods may prove to be costly and time-consuming. More research and advancement is necessary to conclusively select a superior biopsy. When decisive which biopsy to use, the most likely deciding factor is which phase of the process a patient is in: screening, diagnosis, treatment, remission, or relapse. From there, the best decision can be made.
BioChain: Ease and Convenience
BioChain is proud to offer a range of specialized products designed to extract cfDNA in blood plasma or serum. The magnetic bead-based product can perform with less than 1 mL of a sample due maximizing sensitivity of cfDNA. In fact, BioChain’s kit recovers more cfDNA per mL of plasma when compared to competing products, possesses convenient automation compatibility, and is strictly tested for consistency in results.
The extraction kit yields results for Next Generation Sequencing, and the scalable system allows the sample to be conserved and used only as needed. The significantly cost-effective method can be utilized for further applications, from prenatal genetic testing to early cancer diagnoses. Compared to traditional biopsy methods, BioChain’s cfDNA extraction kits can provide a more efficient method of extraction from smaller fragmented chains.
The vamPure Blood Kit also provides a method of DNA extraction from whole blood samples. The advantage of this kit is that the buffer used is free from EDTA for specific experiments. EDTA can be added separately for long-term storage. This kit also supports automation.
As well as the kits, BioChain independently provides a PCR Purification Kit, which increases the quality of a DNA sequence by removing enzymes, salts, buffers, and short DNA fragments. This DNA amplification kit is compatible with automation methods, saving time and costs. Leading at the forefront of biotechnological advancement, BioChain continues to couple premier therapeutic products with competitive pricing to provide an exceptional clinical and research experience.
References
- Harris, Richard. When Genetic Tests Disagree About Best Option For Cancer Treatment. NPR Science Desk. Web. Mar. 2018.
- Gen News. Liquid Biopsy Predicts Colon Cancer Recurrence. Genetic Engineering and Biotechnology News. Web. Mar. 2018.
- Kuderer, N, et al. Comparison of 2 Commercially Available Next-Generation Sequencing Platforms in Oncology. JAMA Oncology. 2017;3(7):996–998. doi:10.1001/jamaoncol.2016.4983. Mar. 2018.
- Mone, Amy. Liquid Biopsy Results Differed Substantially Between Two Providers. John Hopkins Medicine. Web. Mar. 2018.
- Parnell, T. When Tests Collide: Perspectives on Liquid vs. Tissue Biopsies. MD News. Web. Mar. 2018.
- Shtivelman, E. Testing for Tumor Mutations: Liquid Biopsy Versus Traditional Biopsy. Cancer Commons. Web. Mar. 2018.
- Zhang, Sarah. One Patient, Two Cancer DNA Tests, Two Different Results. The Atlantic Monthly Group. 2016. Web. Mar. 2018.
Author
BioChain Institute Inc.

