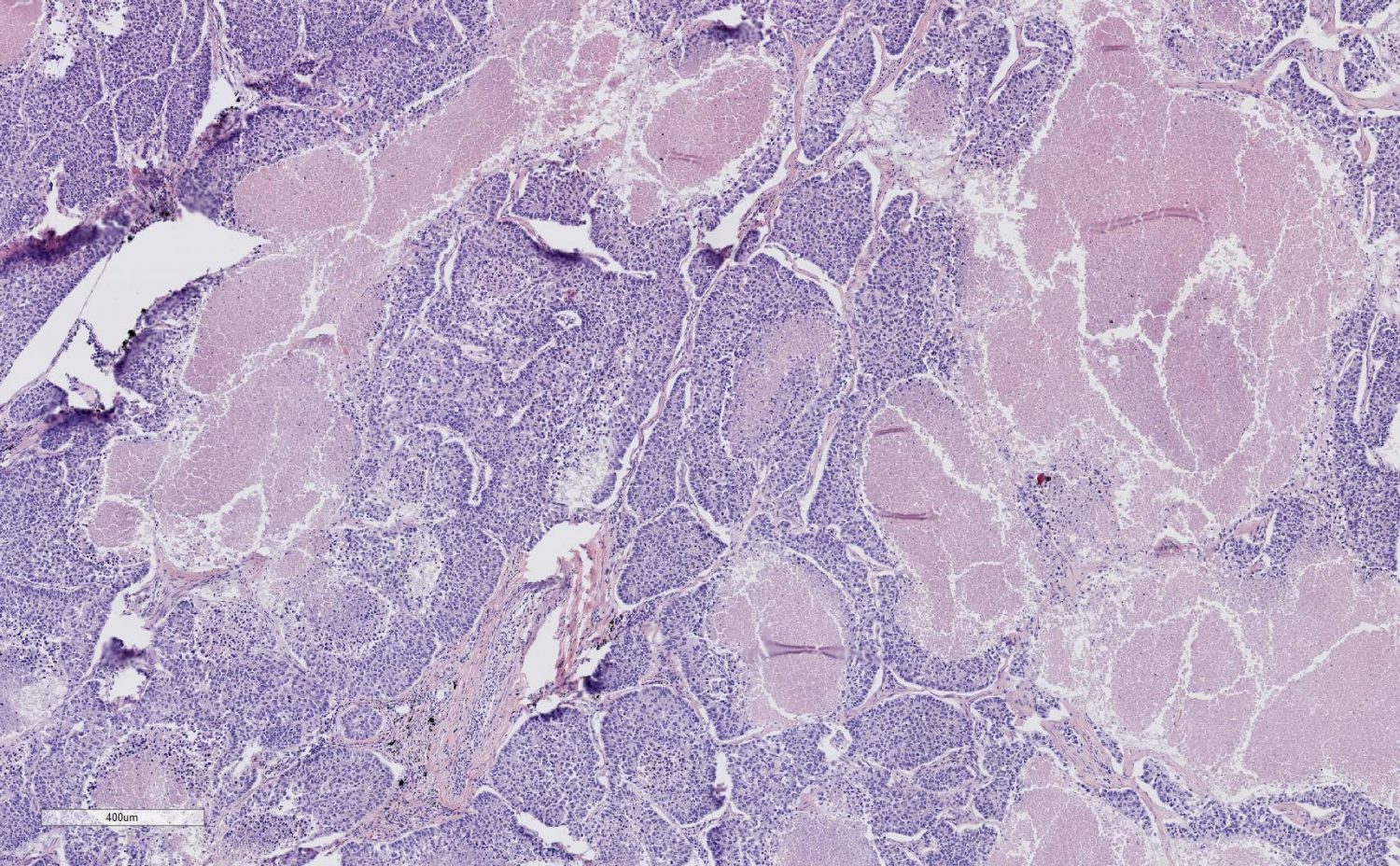
FFPE DNA Damage
Formalin-fixedparaffin-embedded (FFPE) tissues are a very common source of archived tissue samples that have been used by pathologists for decades. It allows for a long-term, room temperature storage solution for tissue samples, making it very convenient. Having been the standard for such a long time, it is also the predominant source of biological research materials. Though it is great for preserving the tissue structures, it can also damage the nucleic acids present. In DNA, depurination or depyrimidation can occur. This is due to hydrolysis of the glycosidic bond, releasing the base, and can become more prevalent with a lower pH. Deamination can also transpire, primarily on the cytosine base to form uracil.
When speaking of DNA damage events, it should not be surprising that guanine will be oxidized to 8-hydroxyguanine, but there is also oxidation of thymine and cytosine bases to form glycols. Other damage events can also occur, such as dimers, nicks, and double strand breaks. To combat this problem, there have been kits developed to repair FFPE DNA damage. These include a mix of enzymes that can mend deamination of cytosine, oxidized bases, nicks and gaps.

Ways to Extract FFPE DNA
Though FFPE tissues do result in damaged DNA, it is still a large source of biological material to study and methods for extracting DNA have improved. Currently, the four main techniques used to extract FFPE DNA are; Proteinase K digestion, which digests the proteins present in the sample and frees DNA that were cross-linked to them; heat treatment, which melts the paraffin, lyses cells and decreases cross-linking; silica membrane based column extraction, which lyses cells and uses spin columns to purify small amounts of DNA; and magnetic silica beads based extraction, which lyses the cell and is incubated with magnetic beads to extract the DNA. Most of these methods would also require purification of the collected DNA before it could be used.
Kits have also been developed, making it easier to perform. The main principal in which they work, like the previous methods, is to de-crosslink the DNA. Many follow the same general steps of removing the paraffin with xylene or a non-volatile, non-toxic reagent, lysing the cell using heat or buffer, digesting the proteins primarily with proteinase K, and extracting the DNA with magnetic beads or simply using then without further purification.
If one is making a fresh FFPE tissue sample and planning to study the DNA inside, here are some tips to improve both the quality and yield of DNA extracted:
- The tissues themselves should be fixed within an hour, and the optimal fixation time is between 12 and 24 hours with formalin that is at a neutral pH. It should then be completely dried before embedding.
- The storage of blocks without cut faces prevents unnecessary exposure to the air and other environmental factors that could damage the DNA.
- The tissue thickness should be between 10 and 20 µm with a surface area between 50 and 300 mm2.
- Extra paraffin should be removed before any purification.

FFPE Tissue Array
FFPE Tissue Panel

FFPE Tissue Section
BioChain: Ease and Convenience
BioChain delivers a comprehensive selection of exceptional DNA isolation kits and reagents to help maximize the confidence that you have in your results. We provide column based kits for manual and magnetic bead based for automated extractions. Our extraction kits can be used for FFPE or Frozen tissue and bodily fluids (e.g. blood, plasma, urine, saliva, and stool). BioChain is also the leading provider of gDNAs and FFPE DNA that have been extracted from hundreds of different tissues and cells. Bulk quantities and OEM manufacturing are also available for purchase at highly competitive prices.

Author
BioChain Institute Inc.

