
What is ctDNA?
Circulating tumor DNA (ctDNA) originates from tumor cells. It is a part of cell-free DNA that is distinguishable by characteristics present in the parent tumor, such as methylation mutations, copy number variations, and presence of specific biomarkers. Increasingly, ctDNA has been used in liquid biopsies to diagnose, treat, and monitor cancer. Find more details on ctDNA and how it is used in oncology here.
While liquid biopsies using ctDNA has distinct advantages over traditional biopsy methods, such as being noninvasive and cost-effective, it also has certain limitations.
Figure 1: Three theorized methods of ctDNA origination from tumors.
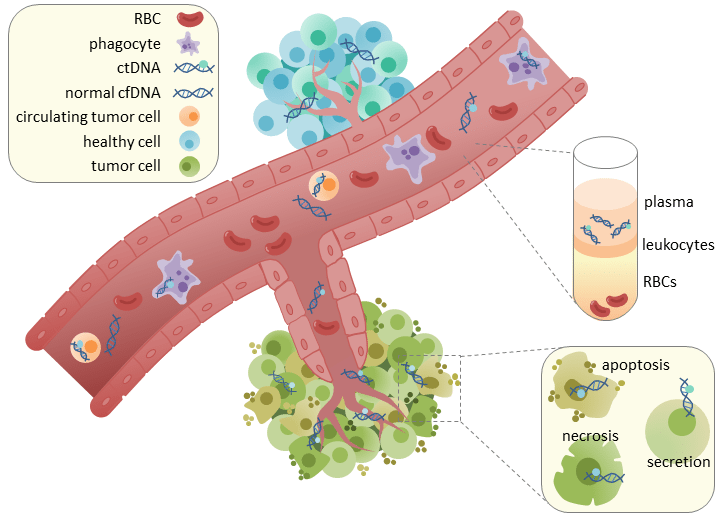
Limitations of ctDNA Liquid Biopsies
False Negatives. This occurs when a liquid biopsy test indicates that ctDNA is not present when it actually is. There are many reasons a false negative can occur, such as low signal-to-noise ratio, and ctDNA’s short half-life. Furthermore, false negatives may also cause researchers or clinicians to miss out on biomarkers of significance.
False Positives. When a liquid biopsy indicates that cancer is present when it is not, it is called a false positive. Some causes include that ctDNA may have been in the bloodstream from the destruction of a tumor – the body’s defense can successfully catch and eradicate the threat. Other times, the ctDNA could have originated from a benign tumor that does not need to be targeted by treatment.
Low Signal-to-Noise Ratio. An undetectable amount of ctDNA is present early in disease. When a cancer is still benign or just starting to metastasize, ctDNA represents a very small percentage of cell free DNA, perhaps less than .01% of a 5 mL sample of plasma. At such low rates, ctDNA detections will need to improve significantly and be specialized to reduce the chance of error.
Short Half Life. Unfortunately, ctDNA in the bloodstream has an extremely short half-life, ranging from 30 minutes up to 2 hours. This causes a range of problems, from tracking tumor heterogeneity to precision treatment. The methodology for biopsies will need to improve in both cost and time.
Lack of Standardization. Due to how recent and innovative liquid biopsies are, the field does not quite have specific standards across clinics and labs in place yet. Liquid biopsies need more consistency in extraction of ctDNA.
BioChain: cfPure® Cell Free DNA Extraction Kit: Testing for ctDNA
BioChain offers the cfPure® Extraction kit for ctDNA liquid biopsies. This kit is designed for rapid and efficient purification of DNA using silica-coated paramagnetic particles. The system is designed to reduce false positives and false negatives by maximizing recovery of 100 bp to 500 bp DNA fragments. Samples of serum or plasma can be as small as .2 mL up to 10 mL.
The kit also provides a solution to the low signal-to-noise ratio and short half-life of ctDNA by creating a scalable system with competitive pricing that works rapidly within 40 minutes. The automation-friendly protocol allows multiple samples to be processed at the same time. In third party tests done on cfPure® and the competitor’s product, it was found that BioChain’s automation yielded more DNA.
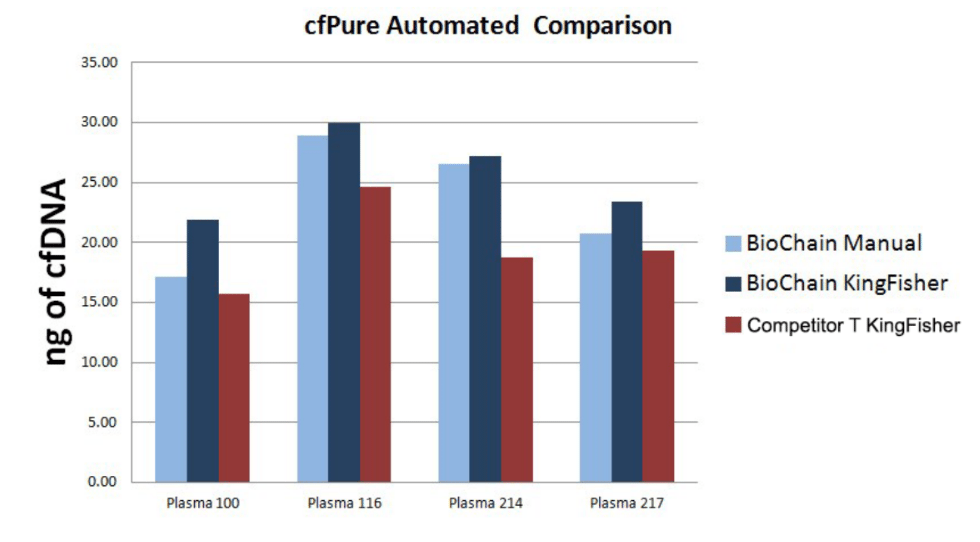
Furthermore, cfPure® aids in standardization by providing consistent reproducible results over time. In multiple experiments done on 1 mL samples of plasma from different donors, total DNA recovery remained the same for each specific donor. The kit is a cost-effective solution to the challenges of using ctDNA in liquid biopsies.
Author
BioChain Institute Inc.

