The Global Pandemic
The current pandemic of COVID-19 caused by SARS-CoV-2 virus has challenged the governments and healthcare systems worldwide with unprecedented social and economic distress. As of 3rd January 2021, 85.4M individuals have been infected worldwide and 1.8M deaths have been reported.
While it caused individuals to isolate, the scientific and medical community around the globe has united in their mission to discover efficient prophylactic and therapeutic approaches.
Clinical Manifestations
As we know by now, COVID-19 clinical manifestations range from asymptomatic to life threatening. About 15% of the patients experience serious illness and require hospitalization while 5% develop critical life-threatening conditions such as septic shock, cardiac and respiratory failures and multiple organ dysfunction (Tancheva et al., 2020).
Lung and Multiple Organ Damage
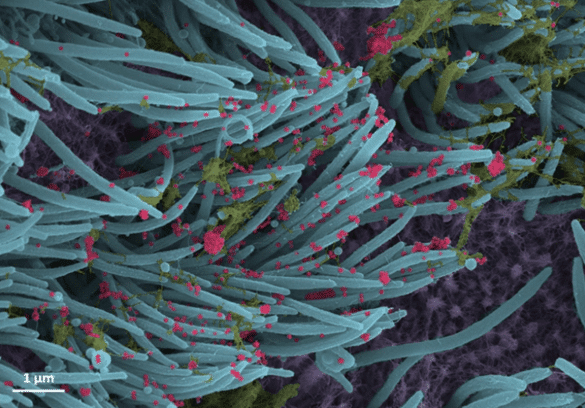
The virus entry and opening in the host cells is facilitated by ACE2 (the human angiotensin-converting enzyme 2) receptor present in most human cells and a ubiquitous protein furin respectively. This explains why SARS-CoV-2 can affect multiple organs including heart, kidney, blood vessels, liver and the central nervous system (Tancheva et al., 2020). In just a couple days, the virus explodes to 10 million from 1000 particles spreading from lungs to other body parts as well as other individuals. Right Source: This colorized scanning electron microscope image shows thousands of coronavirus particles (red) emerging from infected cells (human lung cells – purple, hair like cilia – blue).
Testing and Vaccines
Three types of tests are available to determine active or recent COVID-19 infection.
- Molecular tests are RT-PCR based and considered gold standard for testing active infections.
- Antigen tests are faster but less sensitive and detect the virus surface proteins.
- Antibody/serological tests detect antibodies, in a blood sample, in response to a recent infection.
Virus can be detected in throat swabs, posterior oropharyngeal saliva, nasopharyngeal swabs, sputum and bronchial fluid but has also been detected in intestinal tract and blood samples. Multiple detection methods are suggested to confirm diagnosis to avoid false negatives for individuals with high clinical suspicion such as chest CT scan combined with repeated swab tests (Hu et al., 2020).
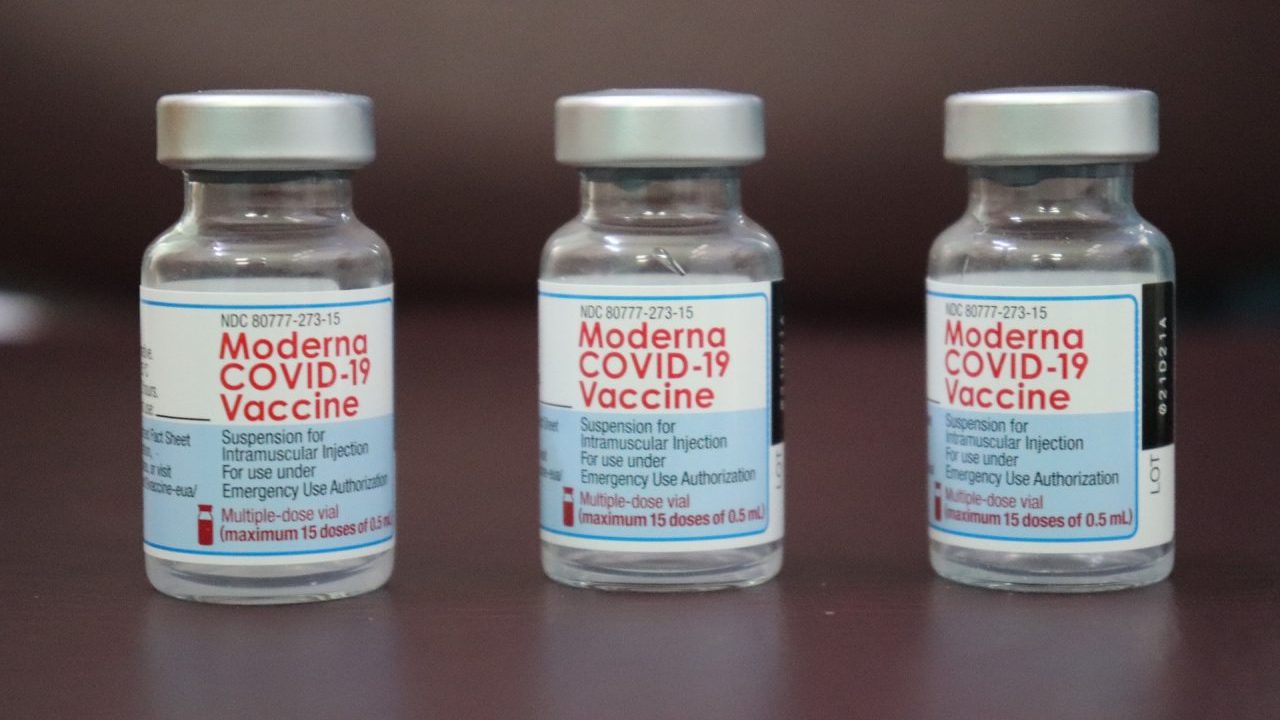
Moderna and Pfizer, became the two leading companies to get authorization and have shown 95% efficacy in phase III trials of their mRNA type vaccine. Their vaccine is an mRNA encapsulated in lipid nanoparticle and encodes the virus’s spike protein which triggers the immune system to recognize the virus.
Some other Adenovirus based vaccines in line are from AstraZeneca-University of Oxford (authorized in UK), Johnson & Johnson and Russia’s National Research Center for Epidemiology and Microbiology.
Covid-19 Variants
The new variant B.1.1.7 with 17 mutations, 8 of which occur in its spike protein that binds with human cells during initial stages of infection, has been reported to cause 60% of the recent cases in London and led to country wide lock down. Click here to read more.
Mutations are common in viruses; however, this mutant is particularly worrisome since 2 of its mutations make it more transmissible and efficient in binding with human cells.
BioChain's COVID-19 Response
At BioChain, we offer quality products to facilitate your COVID-19 research and testing. Our vamPure® Viral nucleic acid extraction kit is ideal for fast and efficient virus detection. Its magnetic bead-based protocol is suitable for manual extraction as well as automation systems. Right: vamPure® kit workflow.
Despite the record-breaking time of vaccine development, number of people vaccinated so far is much less than the expected 20 million by the end of December 2020 and we still have a long way to go in combatting COVID-19. BioChain’s quality products for COVID-19 detection and biospecimen collection can enable you to persist in your research to safeguard the future.
References
- Hu, B., Guo, H., Zhou, P., & Shi, Z. L. (2020). Characteristics of SARS-CoV-2 and COVID-19.Nature Reviews Microbiology, 1-14.
- Tancheva, L., Petralia, M. C., Miteva, S., Dragomanova, S., Solak, A., Kalfin, R., ... & Bramanti, A. (2020). Emerging Neurological and Psychobiological Aspects of COVID-19 Infection.Brain Sciences,10(11), 852.
- COVID-19 Testing: What You Need to Know: Wayne UNC: Goldsboro, NC. (n.d.).
- Terry, M. (2020, November 24). Comparing COVID-19 Vaccines: Timelines, Types and Prices.
- Brothers, W. (2020, December 3). A Timeline of COVID-19 Vaccine Development.
- A Close-up of COVID-19 in Lung Cells. (2020, December 15).
Author
Rikita Gakhar is a biotech graduate based in Bay Area, CA.